L’HISTOIRE SCHILLER
Fondé en 1974 par Alfred E. SCHILLER, ce qui a commencé comme une entreprise individuelle dans un quatre pièces est aujourd’hui un groupement d’entreprises prospère comptant plus de 1 000 employés, 30 filiales et un réseau de vente mondial.
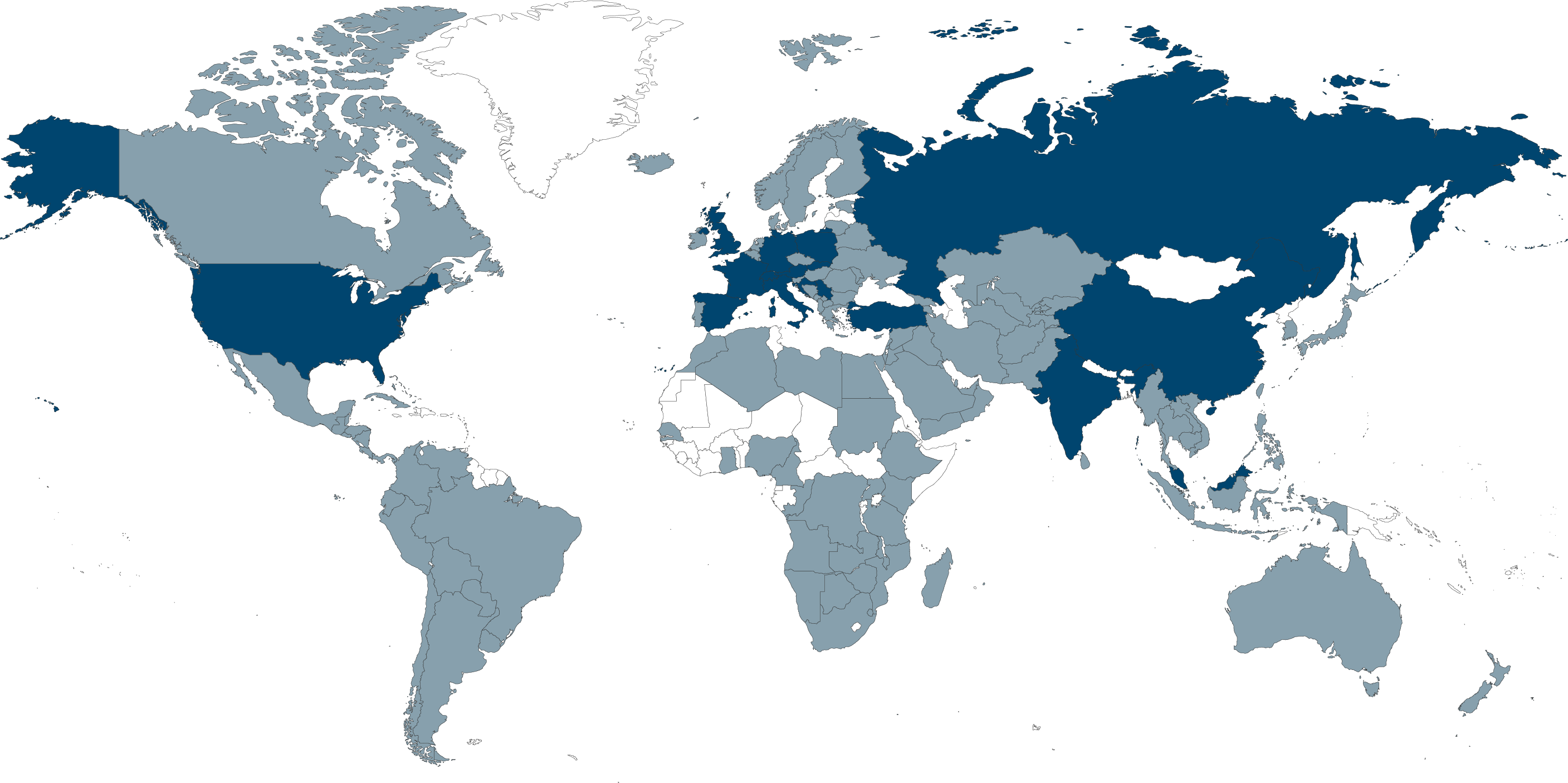
Le groupe SCHILLER est l’un des principaux fabricants et fournisseurs mondiaux d’équipements de diagnostic cardiopulmonaire, de défibrillation et de surveillance des patients, ainsi que de solutions logicielles pour le secteur médical.
Centre d’excellence en défibrillation cardiaque et monitorage patient sous IRM, SCHILLER Médical est établi à Wissembourg, en Alsace. Ce bâtiment accueille les départements R&D, responsables des moniteurs de patients compatibles avec l’IRM et une partie de la ligne d’appareils de défibrillation, ainsi que le site de production de ces deux gammes de produits.
Dirigée par Monsieur David LEOPOLDES, SCHILLER Médical est spécialisée dans la conception, le développement, la fabrication, la vente et le support technique de défibrillateurs et d’appareils de monitorage patient sous IRM.
L’INNOVATION : UNE PRIORITÉ ABSOLUE
SCHILLER Médical ne se repose pas sur ses lauriers – des innovations inédites sont sur le point d’être mises à l’essai.
DES VALEURS FORTES
La satisfaction de nos clients, gage de la pérennité de l’entreprise, et l’application des exigences réglementaires, par un effort permanent de tous pour aller au-delà des attentes.
Produits et services
Engagement et enthousiasme du personnel
Satisfaction client
Innovation constante

SCHILLER s’engage pour la qualité et l’environnement
En complète cohérence avec la politique du développement durable du groupe, la société se mobilise pour agir en industriel responsable et répondre aux attentes des clients.
Nous nous engageons à développer notre Système de Management Environnemental conformément à la norme ISO 14001 :2015 pour nos activités de conception, développement, fabrication, vente et service après-vente de dispositifs médicaux.
 Systèmes de Management environnemental
Systèmes de Management environnemental
Cette certification définit une série d’exigences spécifiques à la mise en place d’un système de management environnemental au sein d’une organisation afin de réduire à court, moyen et long terme tous les impacts environnementaux de l’entreprise (déchets, bruits, énergie)
 Dispositifs médicaux – système de management qualité – exigences a des fins réglementaires
Dispositifs médicaux – système de management qualité – exigences a des fins réglementaires
Certification pour la mise en place de démarches de management qualité pour les fabricants et distributeurs de dispositifs médicaux. Elle défini les exigences d’un système qualité pour les entreprises devant démontrer leur aptitude à fournir des dispositifs médicaux qui soient conformes aux exigences des clients et de la réglementation applicable.
